Researcher Profiles

Christopher B. Hergott, M.D., Ph.D.
2023 Funding recipient
Leveraging neutrophil feedback to combat TET2-mutated clonal hematopoiesis
EvansMDS Young Investigator Award 2023
PROJECT SUMMARY
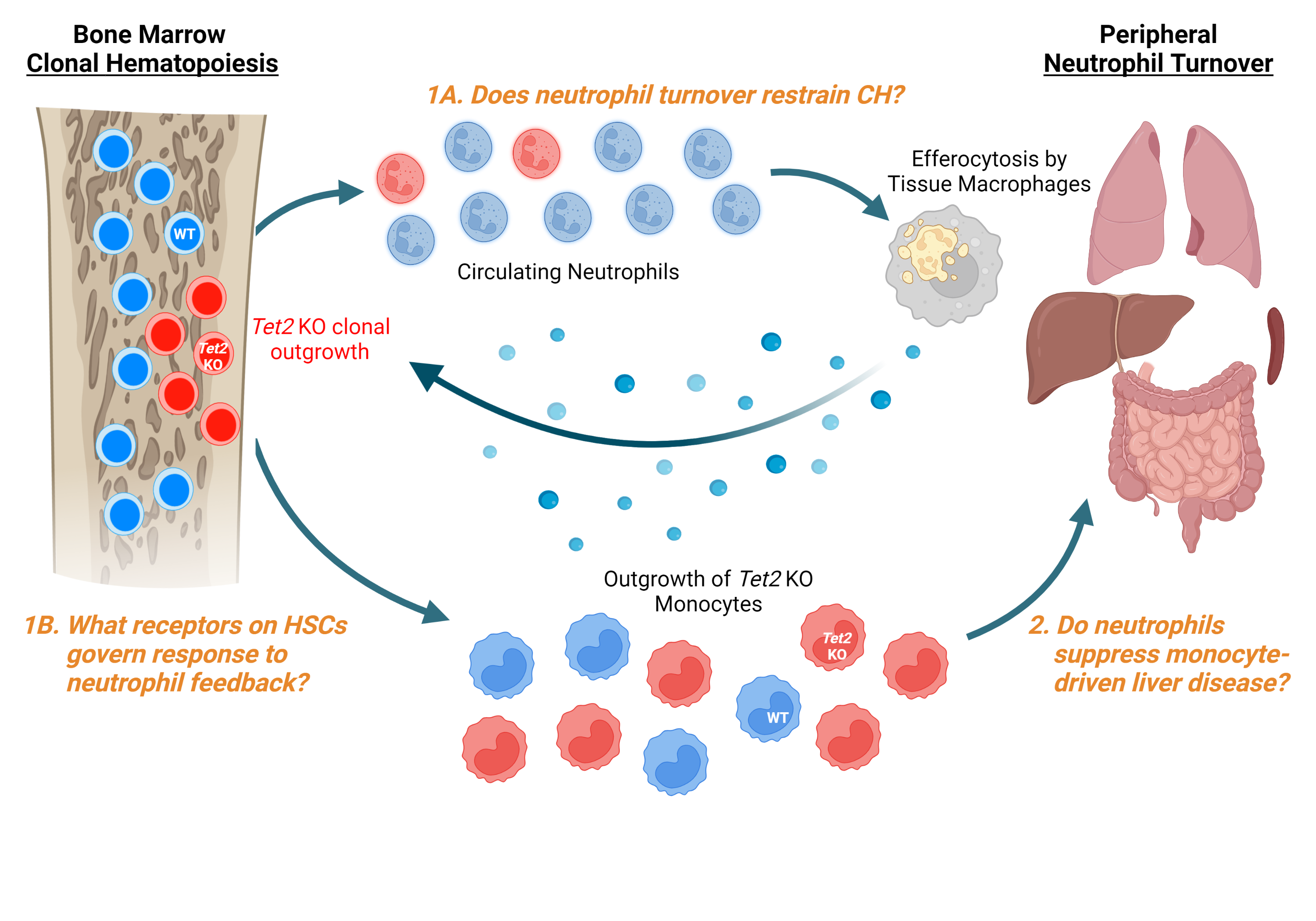
Clonal hematopoiesis (CH) arises from acquired mutations that drive blood-forming stem cells to persist and proliferate over time. More than 10% of people over age 70 harbor at least one of these mutations in their blood. CH produces at least two simultaneous dangers: 1) It serves as a precursor condition for fatal blood cancers like myelodysplastic syndrome (MDS), and 2) It increases the risk of blood clots, chronic liver disease, and other inflammatory disorders by giving rise to abnormally activated immune cells. These hazards have made understanding and restraining CH a top priority in MDS research. However, efforts to combat CH are limited by an incomplete understanding of the forces that regulate its expansion in living hosts, especially regarding the influence of the immune system. In the healthy bone marrow, feedback signals from mature neutrophils (the most abundant circulating immune cell in humans) control multiple aspects of stem cell behavior, including their replication and differentiation. Whether neutrophil feedback significantly affects CH is unknown. However, clinical observations from MDS patients and my preliminary findings in mice suggest that feedback signals from neutrophils slow the progression of CH. In this project, I will use specialized mouse models to dissect the mechanism by which neutrophils restrain CH, identify the receptors on stem cells essential for receiving these signals, and investigate the impact of this feedback cycle on CH-associated inflammatory liver disease. These results could provide a mechanistic explanation for why low neutrophil counts predict disease progression in MDS and illuminate beneficial feedback signals to mimic as new treatments for patients with CH and MDS.