Researcher Profiles

Amy DeZern, M.D., M.H.S.
2020 Funding recipient
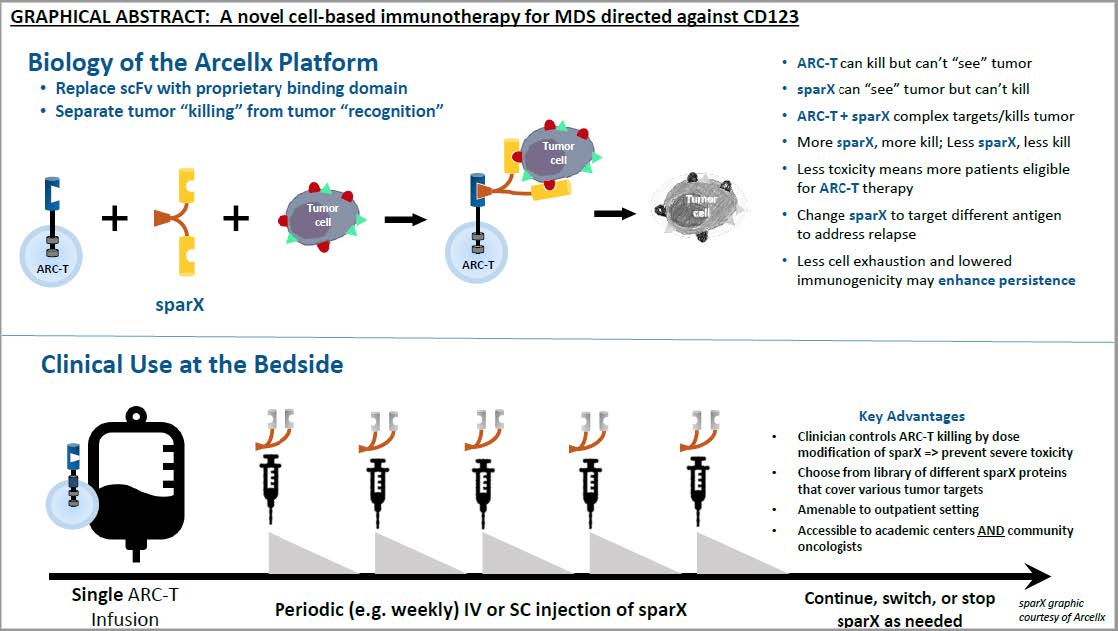
A novel cell-based immunotherapy for the treatment of patients with myelodysplastic syndrome (MDS) directed against CD123
Discovery Research Grant 2020
PROJECT SUMMARY
Myelodysplastic Syndromes (MDS) are a group of bone marrow disorders where abnormal cells replace the bone marrow, causing insufficient production of healthy blood cells. In most cases, MDS eventually results in death and/or evolves to more acute forms of blood cancer such as leukemia, which are also frequently lethal. Current treatment options for MDS patients are very limited and improved therapies are urgently needed.
The focus of our work is to harness the power of the human immune system to fight MDS. To achieve this goal, we have developed a new method that allows us to isolate immune cells (known as “T cells”) from an MDS patient, and to engineer those cells to more effectively target and destroy MDS cells. These immune cells are termed CAR (chimeric antigen receptor) T cells, or CAR T. In the past year, we have focused on improving the efficacy of these CAR T cells using experimental model systems. We demonstrated that our CAR T cell approach could suppress human MDS cells in the laboratory. In the coming year, we will continue to refine this method, and transition the work into human clinical trials.