Researcher Profiles

Alan B. Cantor, M.D., Ph.D.
2021 Funding recipient
Role of ANKRD26 Mutations in Familial MDS Predisposition
Discovery Research Grant 2021
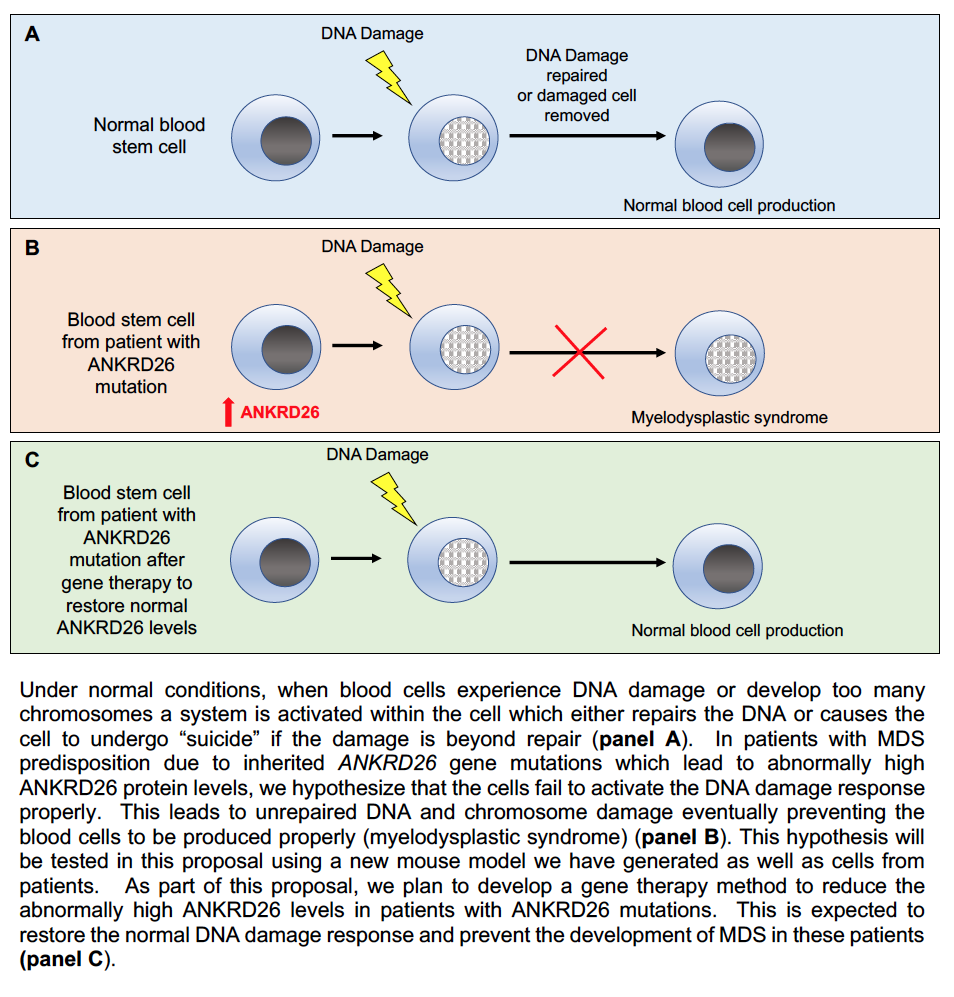
PROJECT SUMMARY
Family members who inherit mutations in the ANKRD26 gene have low platelet counts (cells which help us stop bleeding and increased chance of developing myelodysplastic syndrome (MDS; a disorder of impaired blood cell development) and leukemia (cancer of the blood cells). Yet what ANKRD26 normally does in our bodies and how the mutations cause low platelets and MDS/leukemia predisposition are not well understood. During the 1st year of the project, we further developed a mouse model of this disorder. The results so far suggest that the mice recapitulate a number of features of the disease in humans. This is important because it will allow us to do experiments to elucidate how the mutations cause low platelet counts and increased MDS/leukemia risk in the context of a whole animal (as opposed to cells in a petri dish, which can sometimes provide unreliable results). Moreover, it will provide a system for us to eventually test treatments to improve the platelet counts and lessen the risk of patients developing MDS and leukemia. In separate work over the past year using patient samples, we found that patients with the ANKRD26 mutations may have a problem responding properly to DNA damage. This may help explain why these patients are at elevated risk for developing MDS and leukemia. We have been developing new experimental systems to further investigate this possibility.